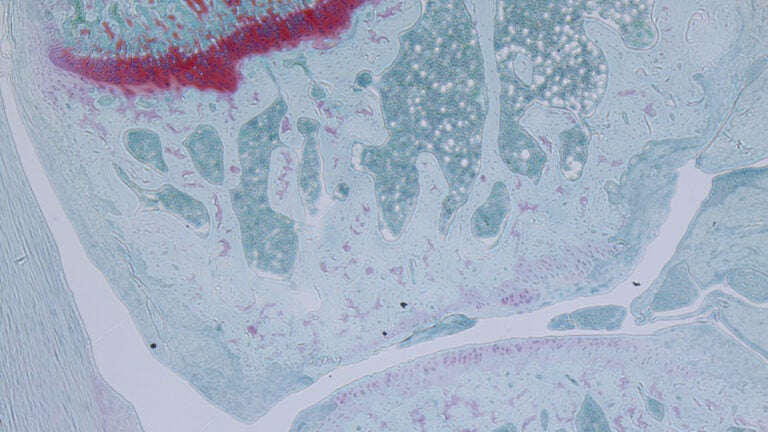
The ability to regenerate joint cartilage cells instead of surgically replacing joints would be a big boon for future patients; image shows microscopic view of joint cartilage cells. (Photo/Nancy Liu, Denis Evseenko Lab, USC Stem Cell)
Joint replacement surgery for arthritis may not be needed in the future
A new molecule may allow patients to opt for a shot instead of joint replacement surgery in the future
Will there be a time when joint replacement surgery for arthritis could be avoided in favor of a shot? USC Stem Cell scientist Denis Evseenko has reason to be optimistic.
In a new publication in the Annals of Rheumatic Diseases, Evseenko’s team describes the promise of a new molecule named Regulator of Cartilage Growth and Differentiation, or RCGD 423 for short.
Discovered by the Evseenko team, the molecule enhances regeneration while curbing inflammation. When the scientists applied it to joint cartilage cells in the laboratory, these cells proliferated more and died less. When the scientists injected it into the knees of rats with damaged cartilage, the animals could more effectively heal their injuries.
RCGD 423 exerts its effects by communicating with a specific molecule in the body. This molecule, called the gp130 receptor, receives two very different types of signals: those that promote cartilage development in the embryo and those that trigger chronic inflammation in the adult. It amplifies the gp130 receptor’s ability to receive the developmental signals that can stimulate cartilage regeneration, while blocking the inflammatory signals that can lead to cartilage degeneration over the long term.
Given these auspicious early results, the team is already laying the groundwork for a clinical trial to test RCGD 423 or a similar molecule as a treatment for osteoarthritis or juvenile arthritis.
The goal is to make an injectable therapy for an early to moderate level of arthritis.
Denis Evseenko
“The goal is to make an injectable therapy for an early to moderate level of arthritis,” said Evseenko, associate professor of orthopedic surgery and stem cell biology and regenerative medicine at the Keck School of Medicine of USC. “It’s not going to cure arthritis, but it will delay the progression of arthritis to the damaging stages when patients need joint replacements, which account for a million surgeries a year in the U.S.”
Evseenko sees RCGD 423 as a prototype for a new class of anti-inflammatory drugs with a broad range of indications. The lab has already developed several structural analogs of the molecule with varying biological effects and potency. His lab is partnering with scientists at USC and beyond to explore the broader potential of these molecules to treat rheumatoid arthritis, jaw arthritis, lupus, neurological and heart diseases and baldness, as well as to maintain pluripotent stem cells in the laboratory.
Evseenko’s team was led by PhD student Ruzanna Shkhyan, postdoc Ben Van Handel and medical student Jacob Bogdanov.
Co-authors of the study include Siyoung Lee, Mila Scheinberg, Nicholas W. Banks, Sean Limfat, Arthur Chernostrik, Carlos Eduardo Franciozi, Ling Wu, Gabriel B. Ferguson, C. Thomas Vangsness Jr. and Nancy Q. Liu from USC; Yifan Yu from USC and Nanjing Medical University in China; Mohammed Parvez Alam, Varghese John, Ali Nsair, Frank A. Petrigliano, Kanagasabai Vadivel and Paul Bajaj from UCLA; and Liming Wang from Nanjing Medical University.
The research was supported by $1.35 million from the National Institutes of Health (K01AR061415) and the Department of Defense (W81XWH-13-1-0465) and $1.2 million from the California Institute for Regenerative Medicine (RB5-07230).